Why don’t patients and their doctors trust generic drugs? Despite the fact that 84 percent of all prescriptions are dispensed in their generic form, there is still a great deal of skepticism about the quality of these less expensive medications.
Although most people recognize that they can save money on generic drugs, many prefer brand names. In one study, nearly a quarter of the women questioned believed that brand name pills were more effective (Research in Social and Administrative Pharmacy, Nov-Dec, 2012). A different study found that many people perceive generic medicine as less potent and not as good as “the real medicine” (Preventing Chronic Disease, online Aug. 30, 2012).
It is hardly surprising that patients might be reluctant to take medicines they perceive as inferior. What about physicians? A survey of doctors found some startling statistics (Annals of Pharmacotherapy, Jan. 2011). About half said they were concerned about the quality of generic medications. About one fourth responded that they did not like to use generics for themselves or their families. The authors urged the FDA to provide more scientific proof supporting the quality of generics if the agency wants to calm such doubts.
A recent article by FDA officials states that “Patients should have confidence that the generic drugs they are prescribed in the United States can be effectively substituted for the brand product or another generic product” (Clinical Pharmacology & Therapeutics, Oct. 2013). The trouble with this assertion is that the FDA rarely provides any data to support it. The gist of its argument is: trust us; we’re the government and we know what we’re doing.
President Ronald Reagan was fond of quoting a Russian proverb, “Trust but verify.” The FDA says generic drugs are “identical” to their brand name counterparts. The problem is that the feds’ definition of identical doesn’t correspond to the usual understanding of the word.
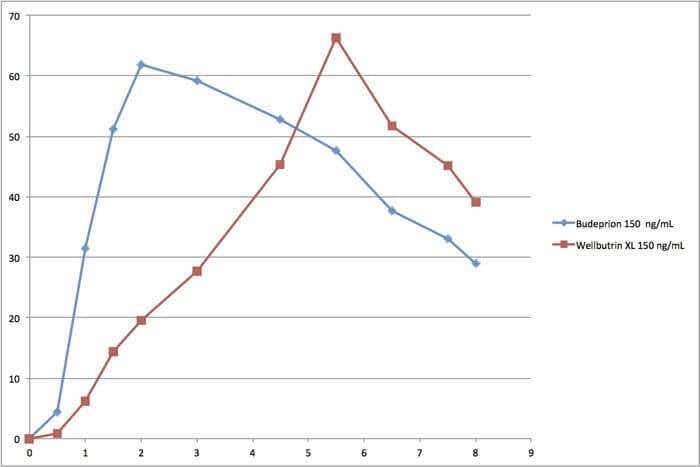
The agency almost never releases data about generic drug bioequivalence–the rate at which the generic drug gets into the blood stream compared to the brand name. In one unusual instance, however, the FDA did make data public. The drug was Budeprion XL 150, which remains on the market as bioequivalent to Wellbutrin XL 150.
At the end of the first hour, the level of the generic drug was more than 400 percent higher than that of the brand name. After two hours, the generic was three times higher. By six hours into the experiment, the brand name had reached a higher blood level and the generic was dropping rapidly. In other words, the generic drug was absorbed and eliminated much more quickly than the brand name, yet the FDA considers these drugs identical. (See the accompanying graph.)
If the FDA wants the American public to trust generic drugs, there are three steps it must take. The first is transparency. The agency should make data on bioequivalence available for verification. The second is by regular inspections of manufacturing plants both here and abroad. The third is continuous monitoring of generic drug quality by random testing of pills from pharmacy shelves. That way, physicians, pharmacists and patients would have more confidence in generic drugs.
No one wants to pay more for medicine unnecessarily. We offer guidelines for using generic drugs safely in our book, Top Screwups Doctors Make and How to Avoid Them.

