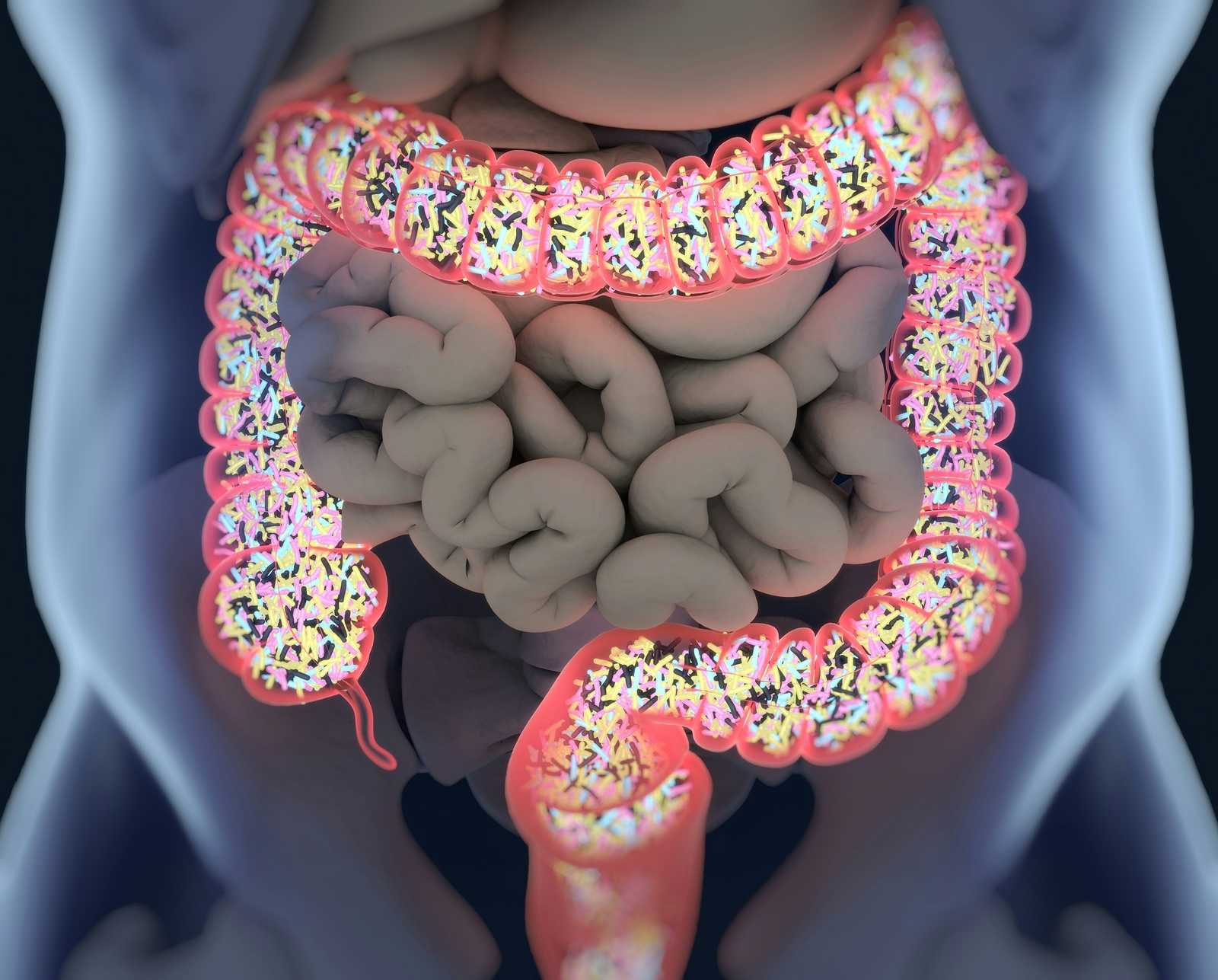
Could you use “we” in referring to yourself? Sure, Queen Elizabeth did that, and probably King Charles does, too. That’s the Royal We. But the rest of us who are not royalty should get used to this idea as well. Science shows that each of us is a multitude, especially when you consider our colon bacteria.
Colon Bacteria and Inflammation:
We are talking, of course, about the microbiome. Over the past few decades, researchers have recognized that we host collections of microbes everywhere on and in our bodies. Colon bacteria (the gut microbiome) came to their attention first. Studying stool samples made it clear that everyone has trillions of microbes in their digestive tracts—not just bacteria, though there are plenty of those. We also host fungi and viruses that feed on the bacteria.
Just how diverse is your microbiome and how much does that matter? One fascinating finding is that each of us has an individual collection of species that is as unique as our fingerprints.
How Do Our Microbes Affect Our Health?
We are accustomed to thinking of colon bacteria as agents of infection. Of course, if you get a rogue strain of E. coli, you may well develop fever, diarrhea and other signs of infection. But even when we don’t have an acute illness, the particular collection of microbes we host seems to influence what diseases we develop.
Inflammatory Bowel Disease and the Microbiome:
Several laboratories have been focused on inflammatory bowel disease (IBD). These autoimmune disorders, which include ulcerative colitis and Crohn’s disease, are devastating. In addition to abdominal pain and diarrhea, patients may also experience weight loss, joint pain, fever and fatigue.
Researchers in Dr. Eran Elinav’s laboratory at the Weizmann Institute of Science in Israel have identified certain types of colon bacteria that are far more common among people with IBD. One of these, a strain of Klebsiella pneumoniae (Kp), triggered symptoms of gut inflammation and diarrhea when administered to previously germ-free mice (Cell, Aug. 4, 2022).
As a result, investigators believe that Kp may lead to IBD. The next phase of this research will be to determine if controlling this germ leads to recovery. The hope is that a collection of viruses (bacteriophages) could offer natural suppression of the bad bacteria. This is a theory best described as the enemy of my enemy is my friend. You can learn much more about this exciting research if you listen to our recent interview with Dr. Elinav. It is Show 1321: Rebalancing Our Microbiome Through Personalized Nutrition and Viruses.
Colon Bacteria and Rheumatoid Arthritis:
Another laboratory has been looking at rheumatoid arthritis (RA) through a similar lens. Researchers at the University of Colorado went looking for the source of autoantibodies that are detectable in people at risk for RA even before symptoms appear. What they found is that these antibodies react to certain gut bacteria, specifically a previously unidentified strain of Subdoligranulum.
When the investigators administered these bacteria to germ-free mice, their paws reddened and swelled in a manner very reminiscent of RA. They also had systemic symptoms. As the scientists note, this specific strain of colon bacteria seems to be driving production of autoantibodies that then attack the joints (Science Translational Medicine, Oct. 26, 2022).
Other Chronic Conditions Linked to Our Microbiome:
Many other chronic ailments may also have their roots in an imbalance of the microbiome. It isn’t just colon bacteria. We also host a vast array of microbes on our skin. Researchers in Japan have recently identified the skin microbe Staphylococcus aureus as a possible bad actor. When it gets out of control, it can trigger a systemic immune reaction ending in lupus (Science Immunology, Oct. 28, 2022).
Discovering ways to restore balance to the various microbial populations in and on our body may someday help us overcome hard-to-treat chronic diseases. Since colon bacteria respond to the food we consume, diet may hold a key.
Citations
- Federici S et al, "Targeted suppression of human IBD-associated gut microbiota commensals by phage consortia for treatment of intestinal inflammation." Cell, Aug. 4, 2022. DOI:https://doi.org/10.1016/j.cell.2022.07.003
- Chriswell ME et al, "Clonal IgA and IgG autoantibodies from individuals at risk for rheumatoid arthritis identify an arthritogenic strain of Subdoligranulum." Science Translational Medicine, Oct. 26, 2022. DOI: 10.1126/scitranslmed.abn516
- Terui H et al, "Staphylococcus aureus skin colonization promotes SLE-like autoimmune inflammation via neutrophil activation and the IL-23/IL-17 axis." Science Immunology, Oct. 28, 2022. DOI: 10.1126/sciimmunol.abm9811

